Matching all aspects of the ISO 15197:2013 performance requirements for blood glucose monitoring systems
Accu-Chek brand of blood glucose monitoring products are compliant with the revised standards of IS/ISO 15197:2013 announced by Central Drugs Standards Control Organisation (CDSCO), Directorate General of Health Services, Ministry of Health & Family Welfare.
What is ISO?
ISO is the International Organization for Standardization headquartered in Switzerland, whose membership is comprised of national standards bodies from approximately 162 countries. ISO is an independent non-governmental organization.
What is an ISO standard?
An ISO standard is a document containing specifications or other precise criteria to be used consistently as rules, guidelines, or definitions of characteristics to ensure that materials, products, processes, and services are able to fulfill their intended use.
What is the ISO 15197 standard?
- The ISO 15197 standard is an international performance standard for blood glucose monitoring (bGM) systems for self-testing in managing diabetes mellitus. This standard:
- Defines requirements that result in acceptable product performance for blood glucose monitoring systems
- Provides guidance on how to demonstrate conformity to the international standard
- The first edition of ISO 15197 was released in 2003: ISO 15197:2003
- In May 2013 the second, revised edition of the standard was published: ISO 15197:2013
- Roche and other manufacturers, health authorities, standards bodies, and clinicians, have been actively engaged in the revision of ISO15197.
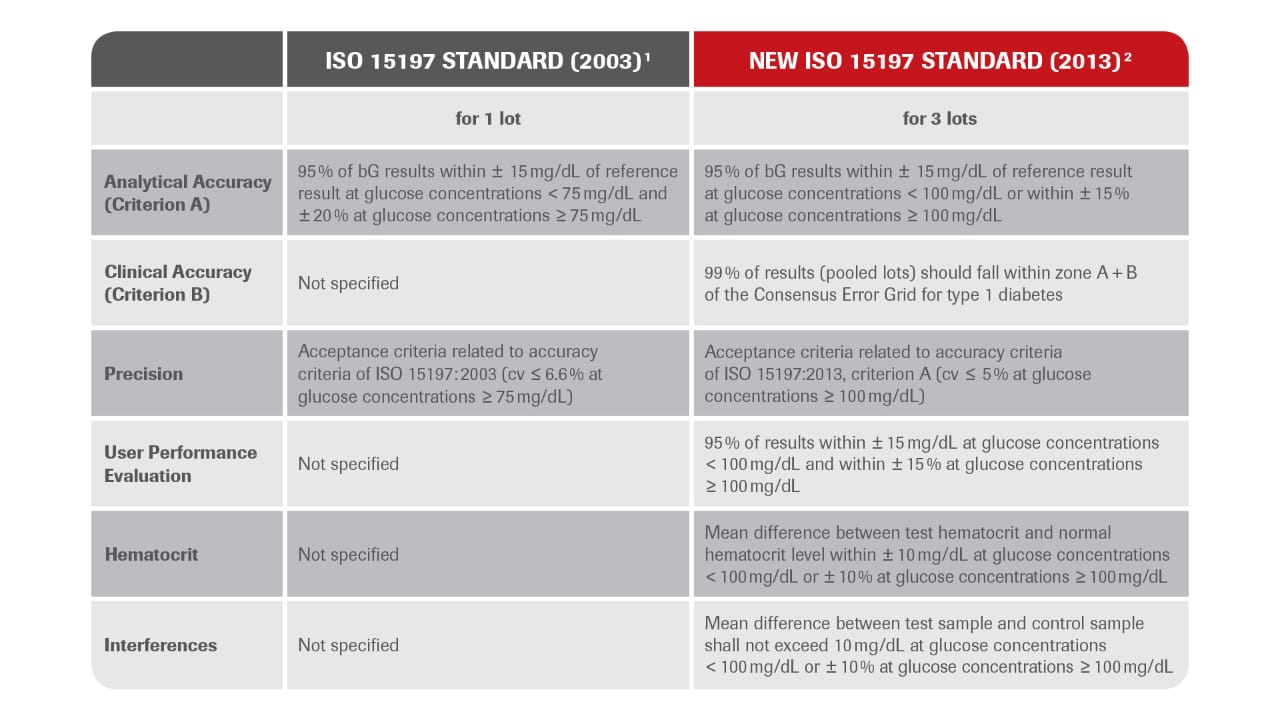
What has changed with ISO 15197: 2013?
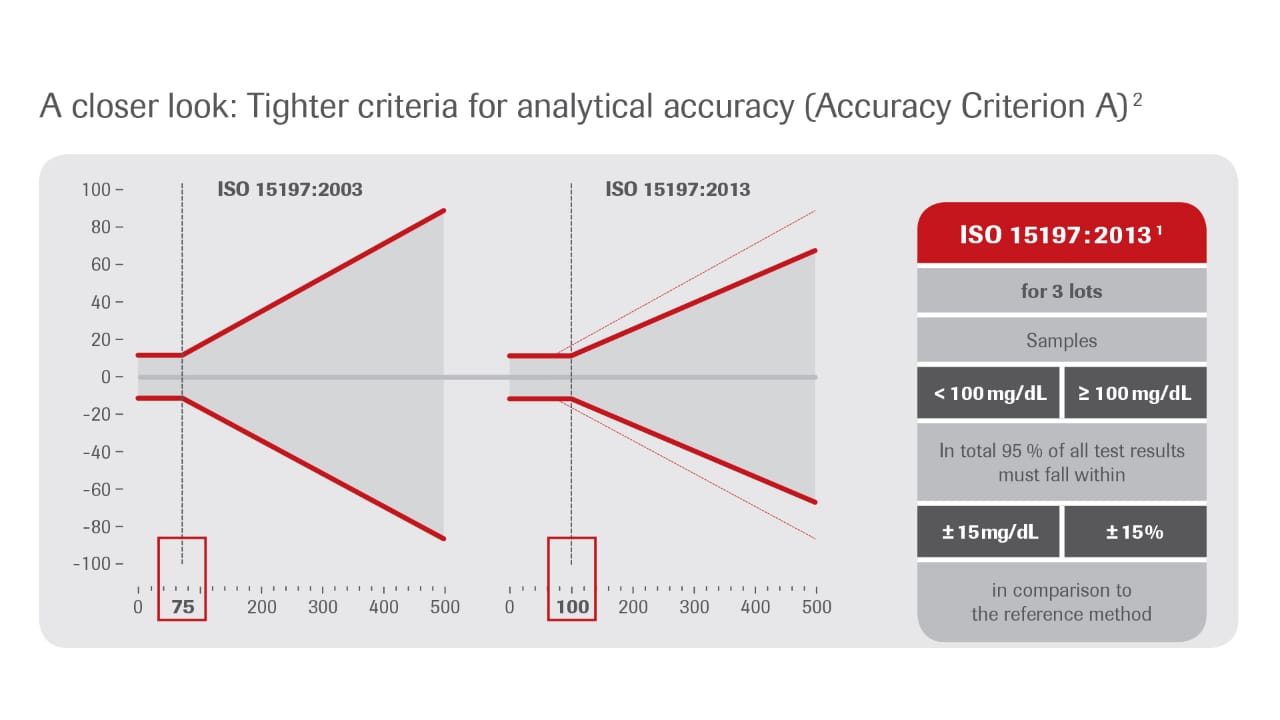
Analytical accuracy (Criterion A): Tighter requirements (so called “15 /15” criteria)
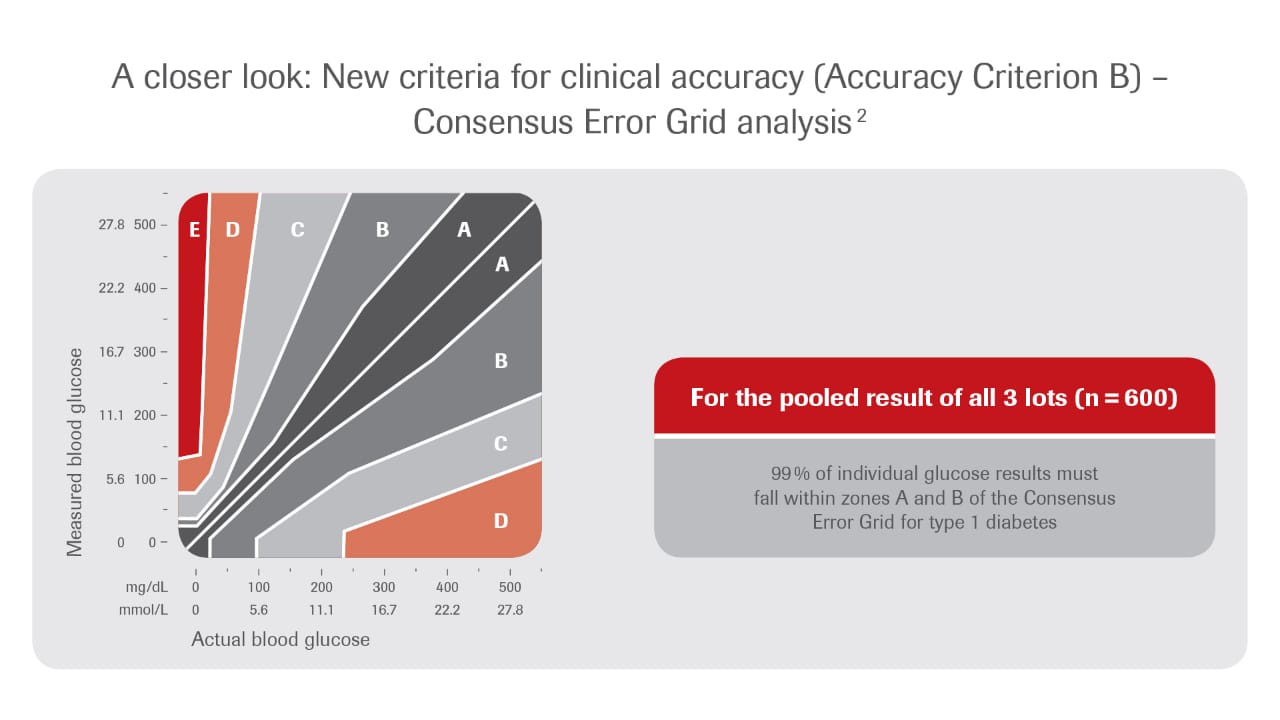
Clinical accuracy (Criterion B): Requirements for a Consensus Error Grid analysis have been specified
Hematocrit and interferences: For the first time acceptance criteria have been specified
User performance evaluation: For the first time acceptance criteria have been specified